FDA approval of adaptive deep brain stimulation (aDBS) within Medtronic’s Percept system was an important milestone in the broader shift towards closed-loop neuromodulation. Now the question is how far adaptivity can go. Nick Collier, CTO at Sagentia Medical, says addressing constraints around computational complexity and power consumption will be key.
One year on from the FDA approval of aDBS in Parkinson’s disease, adaptive neuromodulation has moved from concept to clinical reality. It demonstrates that closed-loop systems capable of adjusting stimulation in real time can meet regulatory expectations and deliver meaningful patient benefit. With several adaptive approaches emerging across neuromodulation, attention is turning to the next phase of development and the technical capabilities required to support it.
The rise of closed-loop neuromodulation
Electrical stimulation of specific nerves or brain regions for neuromodulation therapy is typically delivered via implantable devices combining electrodes, a control unit, and a power source. Historically, most systems have used an open-loop configuration, delivering stimulation using fixed parameters for pulse amplitude, width and frequency. Settings are determined and refined by clinicians, which usually involves multiple patient visits, especially where symptoms fluctuate or the therapy takes time to become effective.
Recent advances in adaptive neuromodulation show how closed-loop systems can respond to changing conditions. In some cases, adaptive control compensates for physical factors such as changes in patient posture. In others, including aDBS, it is driven by physiological feedback that reflects the patient’s response to treatment.
Medtronic’s aDBS is a notable example of the latter approach, using real-time feedback to improve symptom control. It senses physiological signals, interprets their significance, and adjusts stimulation in response.
Further advancements in closed-loop systems could build on these achievements in Parkinson’s disease neuromodulation therapy. They could also benefit people with chronic pain, epilepsy, and other conditions currently under clinical investigation for neuromodulation. But continuous sensing and adaptive control comes at a cost. It increases computational demand – and with it, power consumption – placing new constraints on device lifetime and charging requirements.
Future progress will depend in part on the ability to deliver greater on-device intelligence without draining power reserves. When considering stimulation hardware, R&D teams also need to look at the efficiency of sensor strategies, algorithm design, and underlying computational approaches that support the feedback loop.
How feedback affects the power constraint
The sophistication of feedback systems ranges from simple, direct measurement and control to complex model-based approaches.
Advantages of direct feedback are that it brings minimal computational overhead and if the sampling frequency is low, power consumption is also low. Well suited to resource-constrained hardware, it is currently found on neuromodulation device implants for several conditions. Medtronic’s aDBS is an example of a direct feedback process.
More computationally intensive approaches, such as indirect or composite feedback, would enable more robust and context-aware therapy based on multiple signals. And, at the most advanced end of the spectrum, model-based and predictive approaches offer ways to anticipate physiological changes, not simply react to them. Proactive measures to optimise stimulation and reduce latency could open the door to more personalised and durable therapeutic outcomes. However, as physiological signals become more intricate and control objectives more demanding, the computational burden and power requirements increase.
Neuromorphic computing as an alternative to ‘always on’ sensing and control
Neuromorphic computing – inspired by how the brain processes information – is being explored as one potential route to enable real-time, energy-efficient operation. It uses asynchronous, parallel processing to support fast inference and low latency without relying on the high clock speeds associated with conventional processors. Rather than continuously sampling and processing data, neuromorphic approaches activate computation only when relevant neural events occur, enabling richer feedback and inference without a proportional increase in power consumption.

Importantly, this does not represent a departure from clinically validated adaptive strategies, but could form a potential extension of them, addressing architectural limitations that emerge as systems move beyond conventional closed loop feedback. For conditions like epilepsy where signal dynamics are complex and involve different regions and networks of the brain, this holds game-changing potential.
Neuromorphic computing has been explored for decades as an alternative to conventional computer architectures. It involves processing information in an event-driven manner, emphasising energy efficiency, parallel processing and co-location of memory and processing. Much of the research has focused on supercomputing and modelling the human brain, but several commercial solutions aimed at mobile devices or edge processing have recently emerged. These developments are particularly interesting for neuromodulation. The technology may offer ways to enable real-time, energy-efficient operation, allowing implants to deliver adaptive therapy based on continuous monitoring of patient bio-signals.
Hardware implementation options for neuromorphic neuromodulation
The level to which neuromorphic computing lowers the power consumption of neuromodulation varies across different types of hardware implementation.
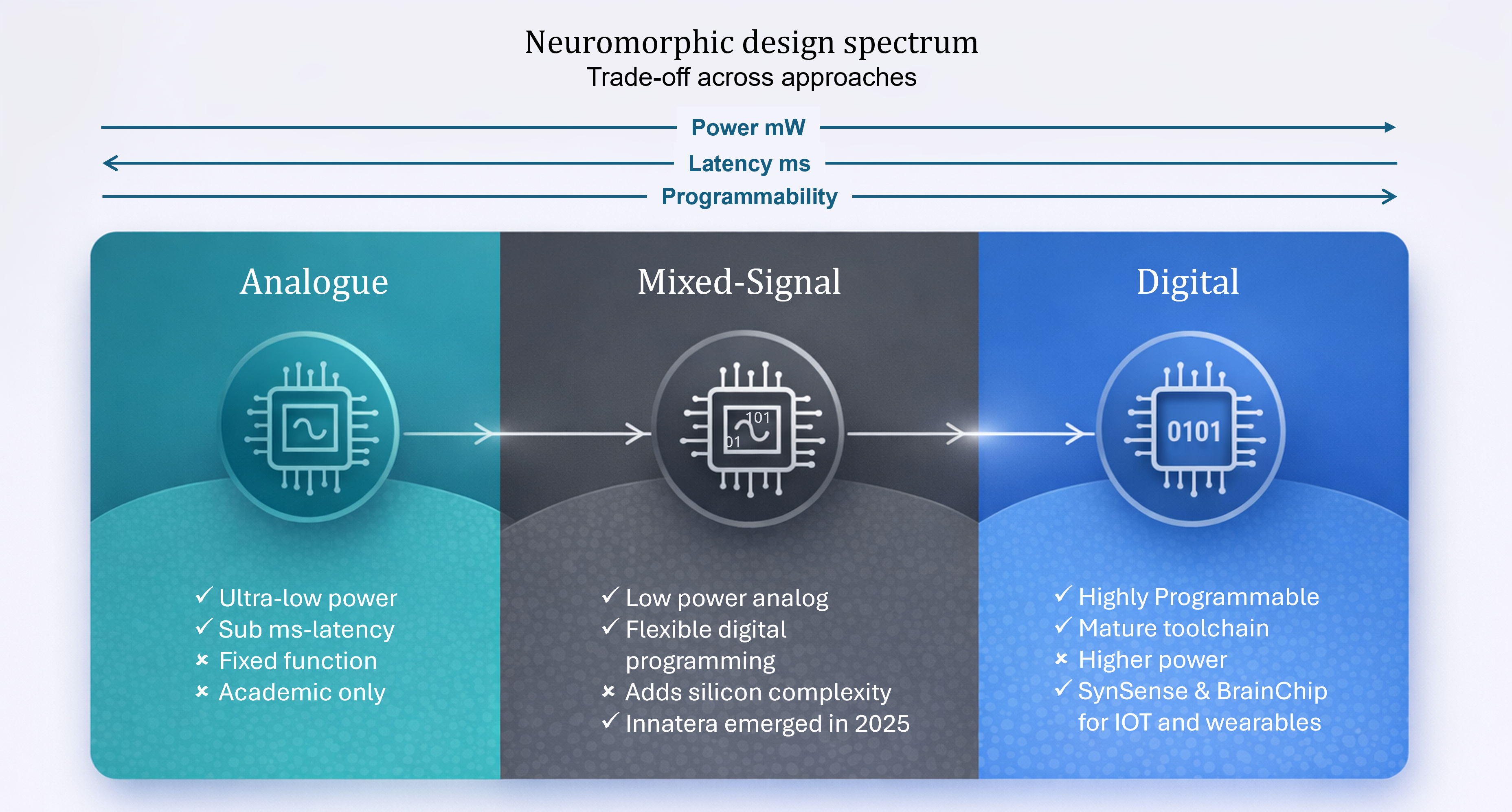
Digital approaches
Digital neuromorphic processors use event-driven computation to process neural signals on standard digital building blocks. By activating computation only when relevant events occur, and keeping memory and computation closely aligned, these architectures reduce unnecessary data movement and power consumption while maintaining predictable behaviour. This improves integration with existing digital systems and makes them suitable for scalable, low-power applications.
Recent developments in these platforms demonstrate that low-power, event-based processing can be realised using mature CMOS fabrication techniques, bringing practical advantages in manufacturability, consistency, and resilience.
However, power requirements remain challenging in the context of implantable devices. Factors such as the use of volatile memory to store synaptic weights, which require periodic refreshing, and the need for analogue-to-digital conversion to synthesise spikes introduce overheads that limit suitability for neuromodulation implants at present.
Analogue approaches
Analogue neuromorphic approaches offer the potential for significantly improved power efficiency.
In these systems, analogue circuits emulate neuronal behaviour in real time, with computation happening only when activity occurs. This can deliver extremely low power operation, which is beneficial in scenarios where energy consumption is tightly constrained.
However, these gains come with trade-offs. Fully analogue designs are inherently less flexible: once chips are fabricated, it’s difficult to modify or refine algorithms, meaning a new hardware iteration is often required if updates are needed. Analogue circuits are also more susceptible to manufacturing variability and noise, which can affect precision and repeatability. Scaling these designs to support larger or more complex networks is a major challenge.
Mixed signal approaches (analogue and digital combined)
Mixed-signal neuromorphic architectures are emerging as a way to balance efficiency with flexibility. They combine analogue neuron and synapse circuits with digital routing, control and programming.
This hybrid approach allows models to be trained or updated in software, then deployed to hardware, which reduces the need for repeated fabrication cycles and enables a single architecture to support multiple applications. In the context of neuromodulation, such designs could allow ultra-low-power continuous monitoring to be handled by analogue circuitry, with higher level control logic invoked only when relevant events are detected.
While mixed-signal neuromorphic technologies are maturing and becoming commercially available, there are still barriers to adoption. The surrounding ecosystem is still developing, with limited software tools and specialised programming models. Manufacturing these hybrid designs is also more complex than producing conventional digital silicon. Right now, they indicate both the promise and the practical challenges of extending neuromorphic principles into safety-critical, implantable medical devices.
Neuromorphic computing in context
Neuromorphic computing illustrates how architectural choices can help address the tension between increased adaptivity and strict energy constraints in neuromodulation systems. However, it represents just one area of exploration. In practice, progress in adaptive neuromodulation will be shaped by a combination of strategies, including more efficient sensing architectures, careful algorithm design, and system-level optimisation of how and when computation is performed. The most effective solutions are likely to emerge from integrated design perspectives rather than a single technology.
Shaping the next phase of adaptive neuromodulation
With closed-loop systems now clinically validated, adaptive neuromodulation is entering a new phase. The challenge has shifted from whether adaptivity is possible to how far it can be extended within the practical constraints of implantable devices.

Addressing this will require system-level design choices that balance therapeutic ambition with what can be delivered safely and efficiently in real-world devices. Future progress will be shaped by how well organisations integrate sensing, algorithms, and architecture into workable solutions.
These are the kinds of decisions and trade-offs being actively discussed across the neuromodulation community. Sagentia Medical will be attending the North American Neuromodulation Society Annual Meeting from 22-25 January, and would welcome the opportunity to connect with others working in this space. If you’d like to discuss strategies for overcoming the constraints facing next-generation devices, get in touch.
Read more of our insights

January 7, 2026
Investable MedTech segments for 2026: growth areas and innovation hotspots


June 20, 2025
Closing the Alzheimer’s gender gap: innovating around early diagnosis and treatment