Existing diagnostic procedures for endometriosis are invasive, have limited diagnostic reliability, and frequently involve long wait times. Here, Sagentia Medical’s Hermione Blakiston and Abigail Collins assess emerging non-invasive methods that are suitable for point-of-care use and could enable earlier treatment. They also explore the standardisation of menstrual blood as a promising diagnostic sample.
Diagnostic challenges of endometriosis
Endometriosis, a complex and painful condition affecting 10% of women (176 million) worldwide, is prone to delayed diagnosis, misdiagnosis, and non-diagnosis. Societal and systemic attitudes that normalise menstrual pain and downplay women’s symptoms can contribute to this, preventing timely investigation and referral.
Obtaining a diagnosis takes almost nine years on average, delaying treatment and risking incorrect treatment or no treatment at all.
Earlier diagnosis could enable targeted clinical intervention to happen sooner. A study published in April 2025, The impact of early diagnosis of endometriosis on quality of life, reports that: “early diagnosis and proper treatment lead to faster relief of patients from pain, and diagnosis of endometriosis at any stage increases the quality of life. Also, early diagnosis decreases complications…like infertility, ureter, and bowel involvement.” Conversely, the absence of accessible, reliable diagnostics can lead to overreliance on empirical treatment which may offer limited clinical benefit.
The gold standard for diagnosis – laparoscopy – involves keyhole surgery. It is invasive and requires specialist skills, which further contributes to diagnostic delay. What’s more, around two thirds of women who undergo laparoscopy do not receive an endometriosis diagnosis, meaning many are exposed to an unnecessary or inconclusive surgical procedure that risks bowel or urologic injury.
Laparoscopy also lacks specificity across different types of endometriosis (i.e., superficial peritoneal, ovarian, deep infiltrating). Visual laparoscopy alone can have specificities as low as ~40% for certain presentations. When used in conjunction with histology to confirm ectopic lesions, there is further diagnostic disconnect. One study found that 38% of laparoscopically diagnosed patients had contradictory histological results, which may be linked to the workflow’s manual, skill-dependent nature.
Imaging methods such as transvaginal ultrasound (TVUS) and MRI are often used ahead of laparoscopy. TVUS is highly accurate for deep infiltrating disease (sensitivity 85-96%, specificity 93-99%), but it rarely detects the more common ovarian or superficial lesions.
Opportunities for non-invasive endometriosis diagnosis
Recent research in minimally- or non-invasive diagnosis of endometriosis centres on three sample types: venous blood, saliva, and menstrual blood. These specimens hold promise for enabling diagnosis at point-of-care – rather than relying solely on invasive procedures in specialist settings – which could improve and accelerate the clinical workflow.
Sample-based diagnostics with specimens collected in the GP office could result in quicker and more effective rule-in / rule-out decisions to enable rapid, targeted clinical interventions such as:
- Immediate justification of hormonal/pain medication treatments
- Informed justification of invasive procedures
- Narrowing down the patient pathology from a broader set of diseases with similar presentation (e.g., uterine fibroids, ovarian cysts, pelvic inflammatory disease, ectopic pregnancy, pelvic adhesions, IBS, interstitial cystitis, colitis, appendicitis)
It is of course important to consider Positive Predictive Value (PPV) and Negative Predictive Value (NPV). Too many false positives would overload the system with unnecessary follow-ups, and false negatives could delay treatment. What’s more, a single test might not cover all endometriosis presentations. Nevertheless, strong performance for the most common subtype (superficial peritoneal) could significantly improve care for many women.
The below chart summarises pros and cons of venous blood, saliva, and menstrual blood in endometriosis diagnostics, as well as start-ups and research groups focusing on the use of these sample types. Urine has also been explored as a non-invasive specimen, but current evidence of diagnostic performance for endometriosis is limited.
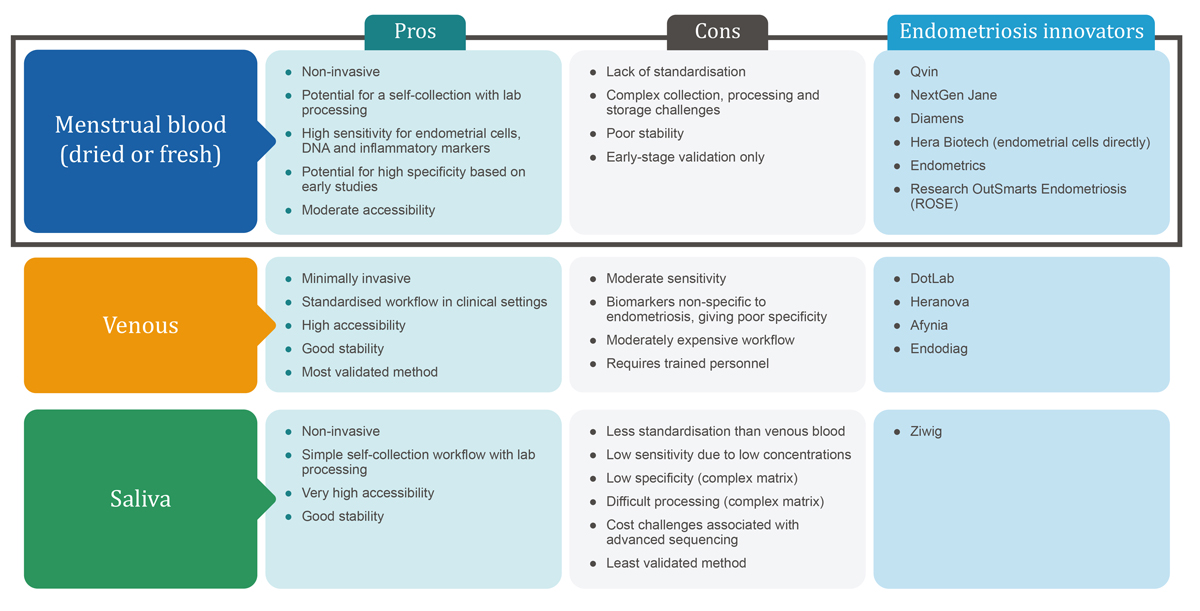 Figure 1: Leading options for sample-based endometriosis diagnosis.
Figure 1: Leading options for sample-based endometriosis diagnosis.
Venous blood is easy to draw in routine care, but it has limited diagnostic value for endometriosis, possibly because it is a systemic sample. For example, sensitivity of serum-based biomarker CA-125 is 38-100% while specificity is 59-99%.
A study focused on saliva as a diagnostic sample reported sensitivity ~97% and specificity ~94-100% for severe ovarian endometriosis. However real-world evidence across all lesion types is limited, and testing up to now has required advanced sequencing and AI analysis.
On the other hand, several studies involving menstrual blood reveal multiple biomarkers (Aromatase, SF-1, HSD17B2 etc.) with reported sensitivity up to 95% and specificity up to 90% across all endometriosis types.
Evidence for the diagnostic performance of saliva and menstrual blood is currently limited to small-scale, single-centre trials. Non-invasive diagnostic tests based on these samples will not replace laparoscopy or imaging without additional validation. However, further exploration of these methods could disrupt and improve the current endometriosis care pathway by reducing the need for surgical referrals or the number of surgical interventions a patient is subjected to. They could also accelerate the surgical referral process for women that need it.
Interventional surgery is only performed for severe disease and is often carried out after a separate investigative procedure. Positive biomarker results could trigger TVUS or MRI for phenotyping and lesion mapping before surgery to reduce the number of operations required. Furthermore, by enabling primary care, these methods could reduce the nine-year gap between symptoms being reported and diagnosis/treatment.
Studies to date indicate that menstrual blood may offer superior diagnostic accuracy to other methods, possibly because it reflects local pathology (e.g. endometrial cells with increased proliferation and migration capacity, immune components, and inflammatory markers). However, this specimen presents several challenges, including the timing of sampling and the collection and processing of samples. Biomarker levels vary widely between women and across the course of a single menstrual period. An individual’s hydration and sample dilution are additional variables, although ratiometric approaches (i.e., normalising biomarker concentrations to a stable internal reference) may mitigate this. Careful biomarker panel selection and standardised sample handling are critical factors for clinical viability.
Despite these challenges, a focus on practical scalability could see menstrual blood adopted as a valuable specimen for enabling earlier diagnosis and treatment of endometriosis.
Menstrual blood as a diagnostic sample
Menstrual blood has not yet been adopted as a clinical sample type, and there is limited historical evidence to support its use. Nevertheless, the Research OutSmarts Endometriosis (ROSE) study has demonstrated the potential of fresh menstrual blood collected via a custom pad-like sponge, revealing stromal cell activity differences through single-cell RNA sequencing. Additional studies suggest that lipid profiles in dried menstrual blood may also serve as viable indicators.
There is growing start-up activity in this space, with a range of collection methods detailed in the summary table below.
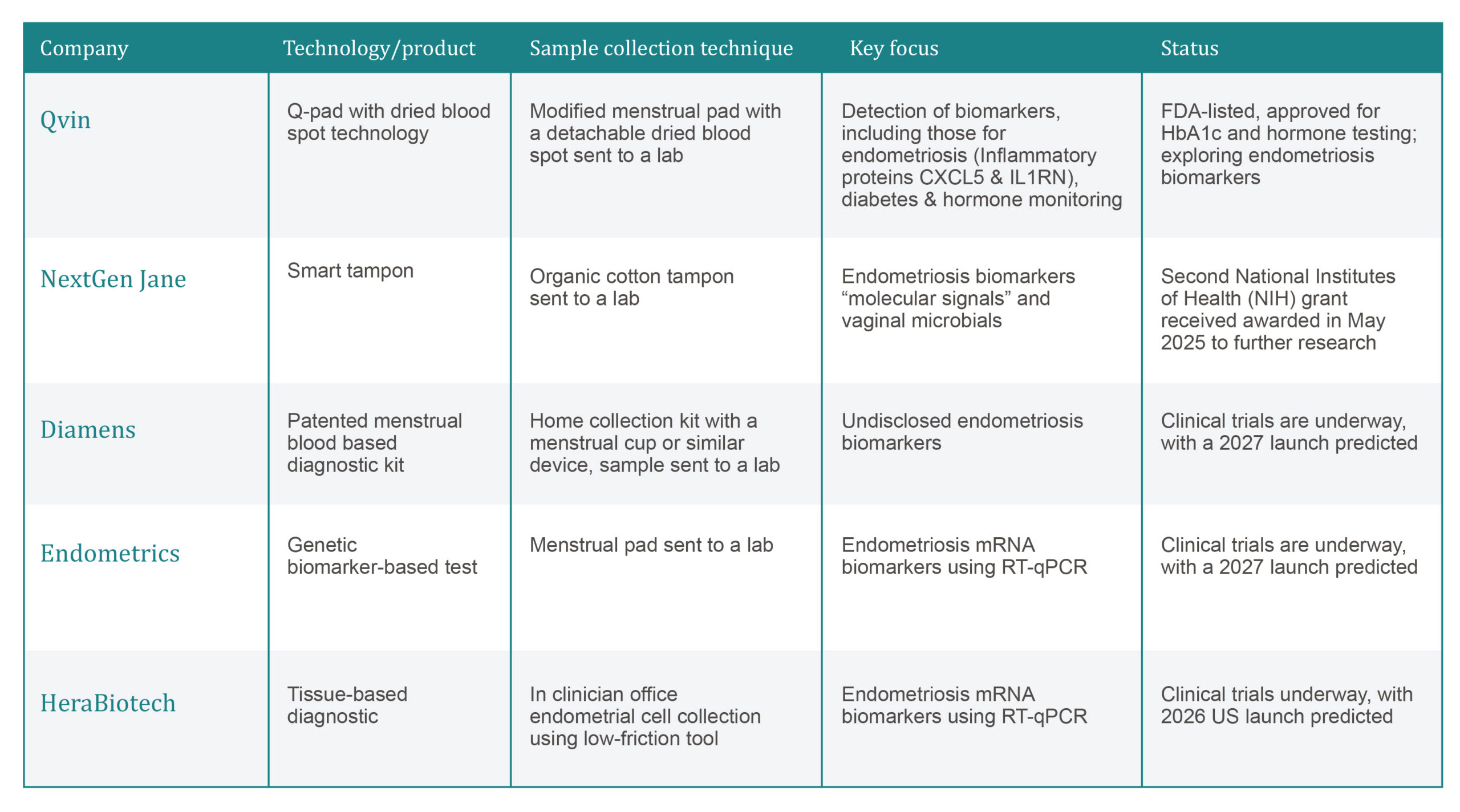
Figure 2: Analysis of start-ups (Qvin, NextGen Jane, Diamens, Endometrics and HeraBiotech) focused on menstrual blood samples for endometriosis diagnosis.
Once safety and reliability are demonstrated from controlled studies and in point-of-care settings, there may be a move to prove equivalence for user-collected samples. This could further enhance the accessibility of diagnosis as well as lowering the associated costs.
Collecting, handling and processing menstrual blood samples
Self-collection of menstrual blood samples sent to a lab or dropped off at a clinic for diagnostic analysis would be game changing. (Read more about self-sampling in this article by Sagentia Medical’s Director of Women’s Health, Kate Marchio). However, the current lack of standardisation in sample collection and storage could be a limiting factor. A core, ongoing goal of The Endometriosis Phenome and Biobanking Harmonisation Project (EPHect) is to develop standard operating procedures for banking biological samples.
Variability in the way samples are handled can be a considerable source of bias and measurement error. At present, start-ups and research organisations use a mix of cups, sponges, pads, tampons, or direct sampling methods.
Absorbent methods are potentially more challenging from a processing perspective; with analysis likely requiring extensive pre-analytical and analytical steps:
Pre-analytical steps (stabilisation and preparation of the sample)
- Dilution with buffer, such as an antibiotic transport medium to prevent microbial growth, or phosphate-buffered saline to facilitate downstream processing
- Reconstitution of the sample into mobile phases for mass spectrometry, flow cytometry or Enzyme-Linked Immunosorbent Assay (ELISA)
- Straining through specialised filters to remove clots, tissue fragments and mucus
- Incubation during red blood cell lysis or enzymatic digestion of tissue-rich samples
Analytical steps (processing and/or measurement of the sample)
- Further incubation to allow antibody staining for analytical techniques or during functional assays
- RBC lysis to access intracellular proteins or eliminate interference
- Centrifugation to pellet nucleated cells, for supernatant extraction
- Freeze-drying of supernatants for solid-phase extraction of proteins
- Washing of non-specifically bound proteins, residual lysis buffer and debris
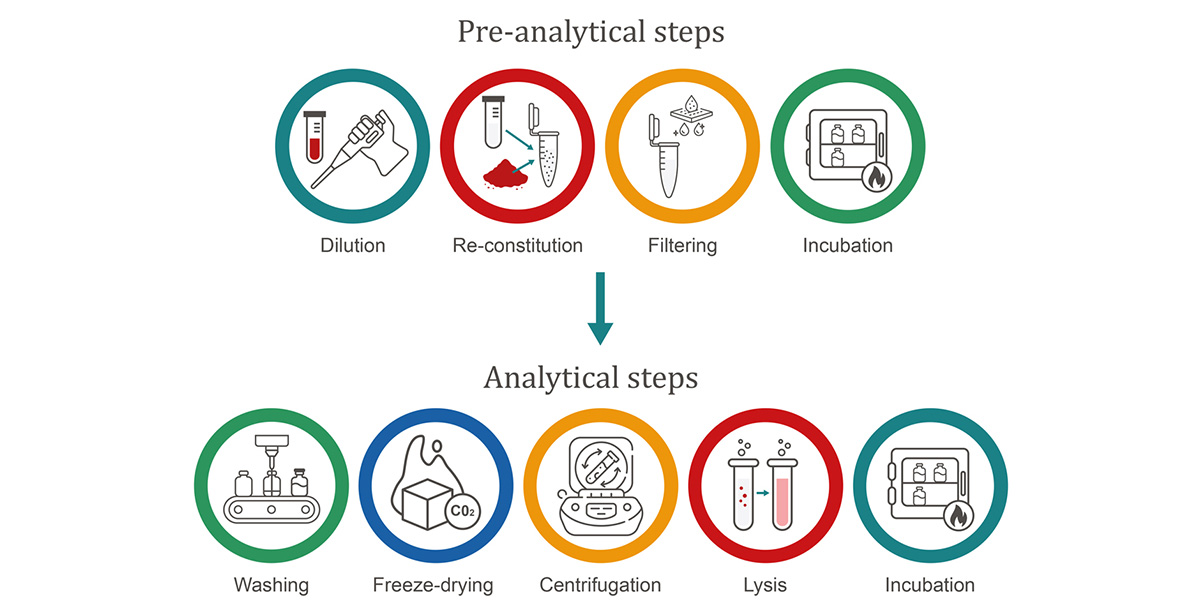
Figure 3: Sample preparation steps.
Theoretical pathways for a lateral flow test
A 2025 study demonstrated an interesting Lateral Flow Test (LFT) solution that could be used for diagnosing endometriosis. It focuses on a borophene nanosheet-based LFT capable of detecting HMGB1 (a relatively stable protein with endometriosis-association) with 500% greater analytical sensitivity than current lab tests.
The study mentions integration into menstrual pads for ‘pregnancy-test-like function’. However, sample collection protocols and the diagnostic pathway are undefined, leaving critical implementation questions unanswered.
To illustrate how an LFT could be applied in practice, we have devised four theoretical use cases and workflows:
- Pre-treated pad: A menstrual pad embedded with filtration materials, buffer salts, and surfactants that condition the blood during wear. After use, the pad – or a detachable section – is sent to a central laboratory for analysis or deposited at a designated collection point, such as a pharmacy or doctor’s office. This approach combines the convenience of at-home sample collection with the delivery of emotionally sensitive results in a clinical setting
- Diagnostic pen: A pen-like device, fitted with a wick that is used to sample blood directly from the vaginal opening. The cap contains a pre-measured buffer reservoir. When the cap is placed onto the test end, a mechanism (such as pierced membrane) releases the buffer, initiating lateral flow across the test pen. This design offers an intuitive workflow with fresh sample collection, similar to the user experience of a pregnancy test
- In-pad removable strip: A wearable pad including a built-in lateral flow strip. After use, the sample pad is removed and the strip is inserted into a lateral flow cassette. A pre-measured buffer is then applied to activate the test. This method reduces the complexity and cost of consumables but involves additional user steps and requires compatibility with a range of sample ages
- Swab-based kit: Inspired by the workflow of rapid COVID-19 tests, this involves swabbing menstrual fluid from a minimally absorbent pad. The swab is then suspended in buffer, and the solution is dispensed onto the LFT for result interpretation. While this method involves more steps, it may benefit from user familiarity and offers a fast, straightforward testing process
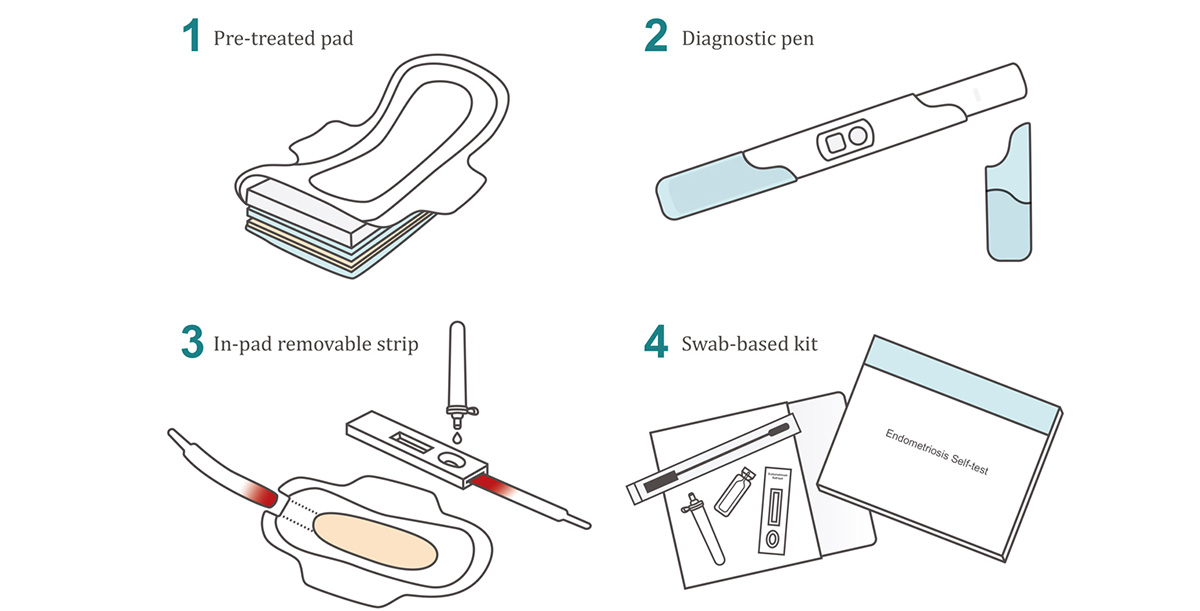
Figure 4: Theoretical use cases of LFT for endometriosis diagnosis.
Balancing diagnostic accessibility and affordability
Progress in menstrual blood diagnostics hinges on establishing a standardised and scalable workflow that delivers superior diagnostic accuracy compared to traditional methods. Human-centred, empathetic design will likely lean towards absorbent approaches due to their simplicity and enablement of self-collection. However, for proof of principle evaluations, protecting biomarker integrity is paramount. So, early MedTech innovation in this space might involve direct aspiration, which requires a higher level of clinical involvement but could still represent a significant improvement on current invasive methods.
In terms of commercial viability, success demands bridging the gap between sample collection and testing. Whether using fresh or dried samples, robust protocols for (self-) sampling, logistics, and preparation are essential. What’s more, as Dr Christine Metz, co-founder of ROSE points out, it’s important to ensure diagnostic tests are ‘reasonably priced’ so as not to be cost-prohibitive once on the market. This applies to the entire end-to-end diagnostic process, from collection to analysis.
In the UK, nearly half of women with endometriosis see a GP 10+ times before it is diagnosed, often undergoing referrals, scans, inappropriate treatments, or emergency visits. With each GP appointment costing the NHS around £49, and laparoscopy or misdiagnosis driving costs into the thousands, a diagnostic price point of £100-£200 per test could undercut the cumulative cost of delayed diagnosis.
Any progress in non-invasive endometriosis diagnostics offers hope for earlier treatment which may shorten the duration of symptoms and control disease progression. And, more specifically, standardisation of menstrual blood as a diagnostic sample could also unlock new clinical pathways for other gynaecological conditions.
How can Sagentia Medical help?
Our science, technology, and engineering consultants have a strong track record innovating around sample collection and preparation, and we’re passionate about translating promising diagnostic science, like menstrual blood-based testing, into real-world impact.
In addition to technical support, we can identify emerging market opportunities, including applications in early cancer detection, PCOS diagnosis, fertility profiling, hormone monitoring and even stem cell re-purposing. Menstrual blood offers an underutilised source of pathological and molecular insights, with potential to support diagnostics across a wide spectrum of conditions.
If you’re looking to integrate a new sample type, such as menstrual blood, into your diagnostic workflow, we’re the ideal innovation partner. Contact us to find out more: [email protected].
Read more of our insights

January 7, 2026
Investable MedTech segments for 2026: growth areas and innovation hotspots


June 20, 2025
Closing the Alzheimer’s gender gap: innovating around early diagnosis and treatment